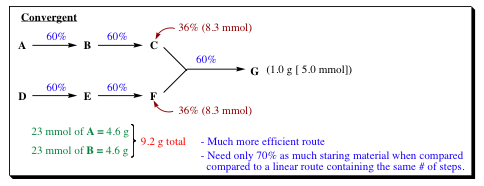
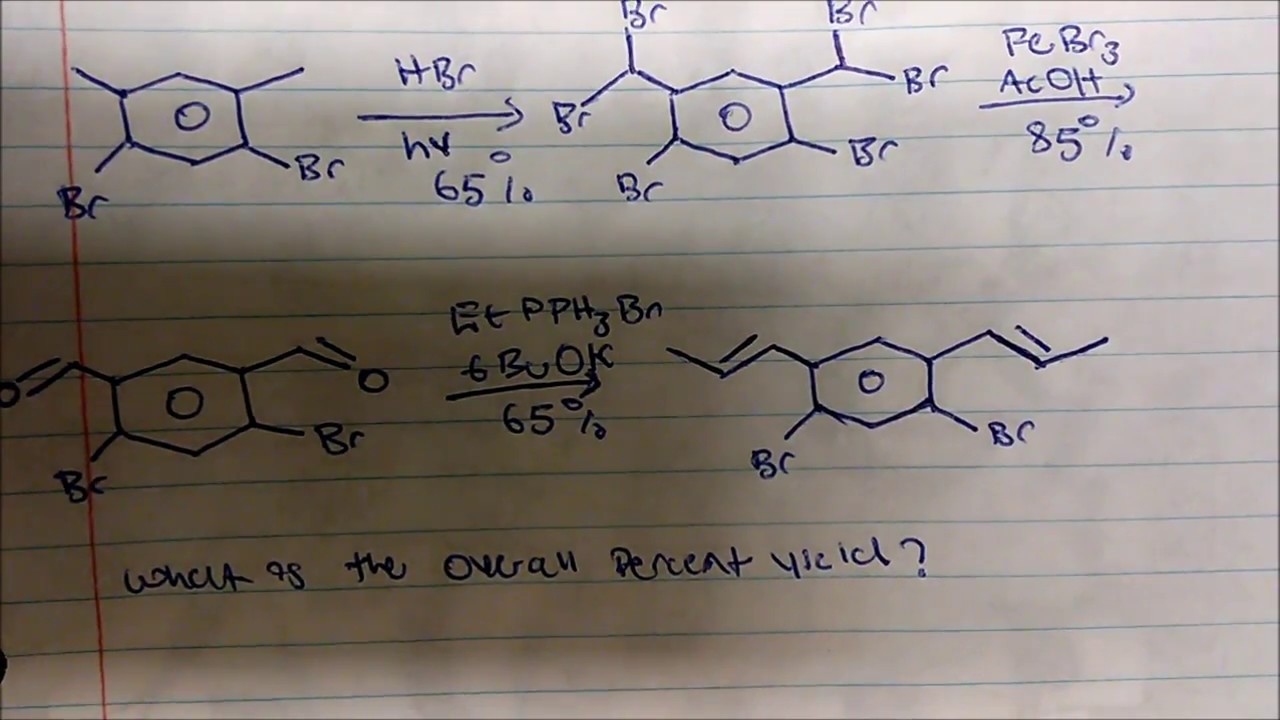
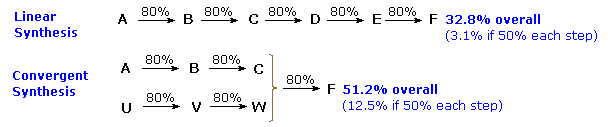
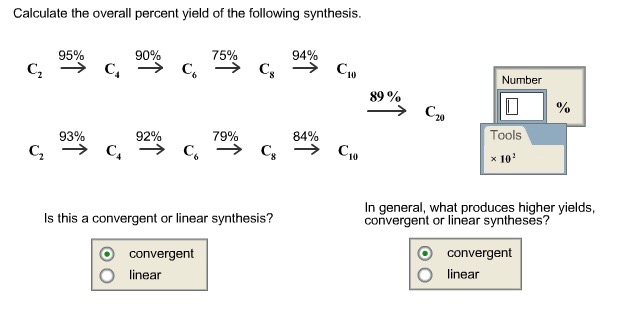
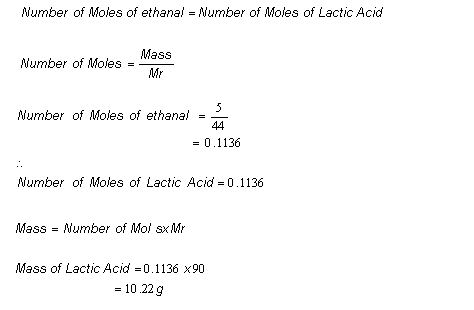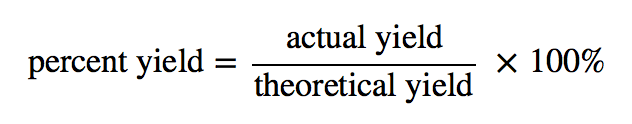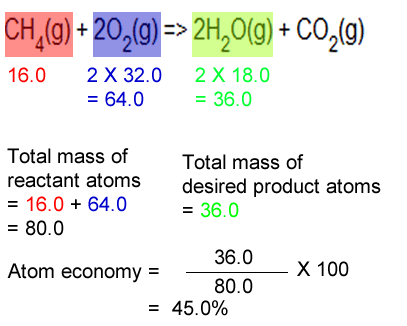Lecture 12 Chemical Reaction Engineering (CRE) is the field that studies the rates and mechanisms of chemical reactions and the design of the reactors. - ppt video online download

Theoretical, Actual, Percent Yield & Error - Limiting Reagent and Excess Reactant That Remains - YouTube

Synthesis of Dihydropyrazoles via Pd‐Catalyzed Heterocyclization/Carbonylation Reaction: Development and Parameterization Studies - Barboza - 2022 - ChemCatChem - Wiley Online Library

reaction mechanism - Do you always need to balance a chemical equation to find the percentage yield and limiting reagent? - Chemistry Stack Exchange

Multigram Synthesis of a Combustion‐Relevant δ‐Ketohydroperoxide through Sulfonylhydrazine Substitution - Ke - 2022 - Chemistry – A European Journal - Wiley Online Library


.jpg)